This essay was written by upper-sixth former Salvatore Nigrelli, and was the winner of the STEM category for the 2020 Independent Learning Assignment. The following provides a short abstract to the full essay, which can be found at the bottom.
Estimated read time of abstract: 4 minutes
Estimated read time of essay: 1 hour 15 minutes
Supramolecular chemistry is all about making functional molecular assemblies without chemically bonding the component molecules together. Take the reaction scheme below:

In this scheme, two molecules are held together only using hydrogen bonds, to form a tennis ball shaped capsule. So it is the art of the supramolecular chemist to try and find innovative ways of making complex assemblies, with only a handful of intermolecular forces at their disposal. This is particularly shown in a type of structure called supramolecular cages.
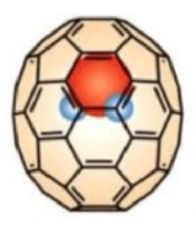
Cages are ubiquitous throughout the world of chemistry. The Buckyball (Figure 2, is a simple type of molecular cage, consisting of 60 carbon atoms in a spherical shell arrangement.

Supramolecular cages take this idea one step further and ask the question: Can we design assemblies that allow us to put a small molecule ‘prisoner’ inside the cage?
Take the Buckyball again. Using a type of complex reaction sequence called a molecular surgery reaction, it is possible to open the Buckyball up and place a water molecule inside, held in by the London forces it can form with the cage walls, transforming the Buckyball into an exciting supramolecular cage (Figure 3).
But you may ask, is there actually any point in making these tiny molecular prisons, or is it purely to indulge a few curious supramolecular chemists? The answer is that, although the field is relatively new, it is becoming paramount that the applications of supramolecular cages are innumerable, from security to chemical analysis, and even cancer therapy.
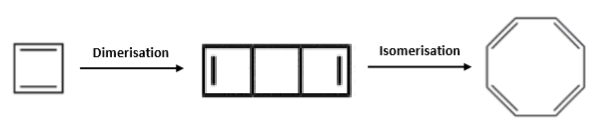
Cyclobutadiene is a pesky, annoying molecule – mainly because it reacts with itself extremely quickly in a dimerisation/isomerisation reaction to produce cyclooctatetraene:
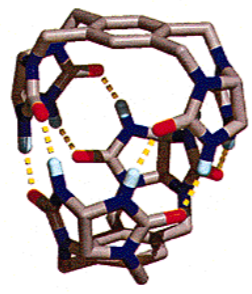
This self-reacting property of cyclobutadiene makes it extremely difficult to probe its chemical structure. Until only a few years ago, the only way that it had been achieved was by holding the molecules in an argon matrix close to absolute zero. However, with the advent of supramolecular cages, all of this changed. If you make a single cyclobutadiene molecule inside a type of supramolecular cage called a carcerand (Figure 5), no other molecules can get to it, so it stays in its original, undimerised form, and can be analysed using NMR spectroscopy. This is a classic example of how supramolecular cages are already revolutionising the field of chemistry.

Figure 5 An Example of a Carcerand
However, not all of the uses of supramolecular cages lie in a lab. The cage below is one such cage with extremely promising applications. In the presence of picric acid molecules, the cage can encapsulate one of them. Once the picric acid is inside the cage, it is close enough for a type of photochemical process called a Förster Resonance Energy Transfer to take place, which causes the cage to completely change colour. This is incredibly useful because picric acid is one of the most common explosives, so cages like these could be used in the next generation of fast, accurate explosives detectors and save countless lives.
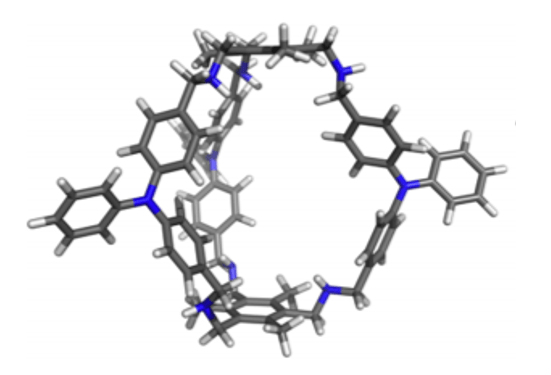
Figure 6 A Supramolecular Cage to be Used in Explosives Detection
Looking into the monumental applications of supramolecular cages got me thinking – could I use the skills in supramolecular chemistry that I picked up over the course of my ILA and design a novel type of supramolecular cage to solve a real world problem? I decided to try and solve the problem of fluorouracil as a chemotherapy drug. The essence of the issue is that fluorouracil is an extremely promising cancer drug, but it also readily attacks brain tissue, and so its use is limited to very extreme cases. This appealed to me as a problem to solve because of the great positive impact that it would have, and the fact that fluorouracil (Figure 7) has a number of structural features that make it very attractive to supramolecular chemists.
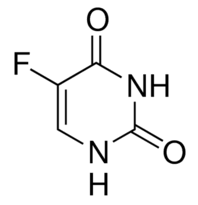
Figure 7 The Structure of Fluorouracil
Fluorouracil can form three strong interactions with its fluorine and two nitrogen atoms that would allow it to be readily encapsulated. So if a cage with three parallel bars could be designed, it would strongly bind the fluorouracil molecule. However, finding a chemical arrangement that allows this to take place proved to be difficult, as it is a rare occurrence in chemistry. In the end, I managed to work out that if the cage used molybdenum centres with thiophene ligands, a trigonal prismatic arrangement around the molybdenum atom would be obtained (Figure 8) , making the three parallel bars possible.
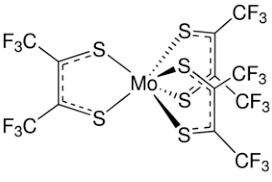
Figure 8 A Molybdenum-Thiophene Complex Showing a Trigonal Prismatic Geometry
The key principle behind the cage is that the fluorouracil molecule stays inside the cage, so it cannot react with anything, until it enters the cancer cells, where the cages are opened, thereby allowing the fluorouracil to kill only the intended cancer cells. This targeted opening is a rather unusual feature of molecules, and so in the end I used the fact that, if nitrophenyl ether groups (Figure 9) were placed on each bar, in the presence of a targeted beam of UV light, the cage could be successfully opened once inside the intended cancer cells.
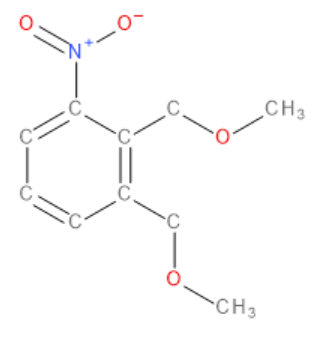
Figure 9 A Nitrophenyl Ether Group
My final cage design is shown below:
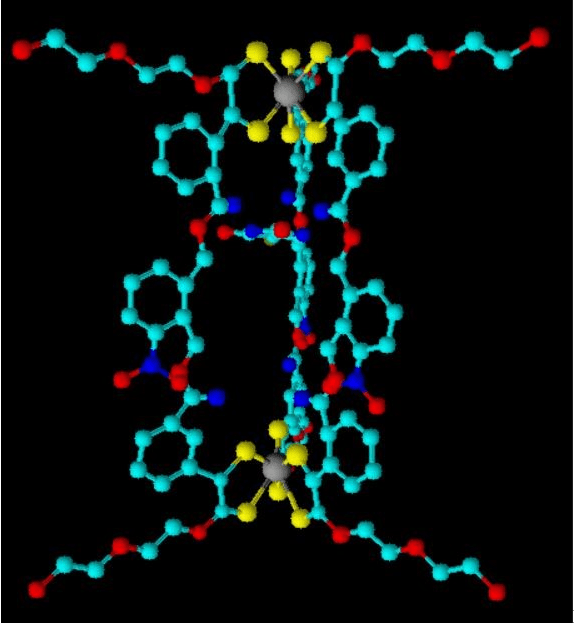
Figure 10 My Final Cage Design
To summarise how my design works: Outside of the body, fluorouracil is encapsulated inside the cage, then a solution containing the encapsulated fluorouracil is injected into the patient’s bloodstream. Whilst inside the cage, no other molecules can get to the fluorouracil so it cannot react with anything and cause its bad side effects. Once the cage reaches the cancerous cells, using UV light, the cages in the cancer cells are opened, releasing the fluorouracil and killing only the intended cells. Therefore, this scheme allows for fluorouracil to be used to treat cancer patients, without causing any negative side effects.
To view Salv’s full article, follow this link below.