This essay was written by upper-sixth former Alex Thow, and a finalist for the 2020 Independent Learning Assignment. The following provides a short abstract to the full essay, which can be found at the bottom.
Estimated read time of abstract: 5 minutes
Estimated read time of essay: 45 minutes
Quantum mechanics is difficult. It is confusing. It is illogical. Albert Einstein himself hated the concept, stating, “If it is correct, it signifies the end of physics as a science”. Erwin Schrödinger, one of the great names in early quantum mechanics, said, “I do not like it, and I am sorry I ever had anything to do with it”. Even Richard Feynman, one of the greatest teachers physics has ever seen, went so far as to say, “I think I can safely say nobody understands quantum mechanics.” Despite all this, it manages to answer some of the most interesting questions out there in a beautiful way. In my ILA I explored the answer to the question of how it is certain molecules have colour.
To begin we must mention molecular orbitals (MOs). Some of the mathematics behind these MOs is discussed in the main text, but unfortunately it is too complicated to go into here, so the results will have to speak for themselves. Electrons in molecules are never in one place – we have quantum mechanics to thank for that. They are spread out unevenly around the molecule and the regions that they occupy are the MOs. There are many MOs in each molecule to accommodate all the electrons, as only two electrons can occupy any given orbital due to an effect called the Pauli exclusion principle. The different distribution of electron density within each MO leads to the MOs having different energies. The highest energy MO with an electron in it is called the HOMO (highest energy occupied molecular orbital) and the lowest energy MO that does not contain an electron is called the LUMO (lowest energy unoccupied molecular orbital). These orbitals are key in so many areas of chemistry, including the chemistry of colour, as it turns out that an electron in the HOMO can absorb the energy in a photon and jump up the LUMO. The energy of a photon of light is directly proportional to the frequency of the light, and so the energy gap between the HOMO and the LUMO determines the colour of light that is absorbed. The colour we see is simply the complementary colour to what is absorbed, as the rest of the spectrum is reflected.
Now, my ILA would have been a lot shorter if it were simple for the HOMO-LUMO energy gap to be in the right range for visible light to be absorbed, but unfortunately this is not the case for most molecules as they generally absorb UV light. The main structural feature that coloured molecules require is something called a conjugated system, which is a chain of alternating double and single bonds (these are generally bonds between carbon atoms as the molecules we are interested in are organic). A clear example of this is in beta-carotene, the molecule that gives carrots their orange colour.
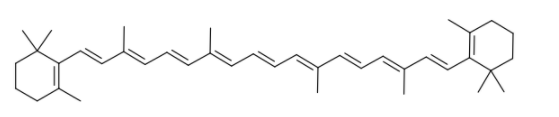
A structure like this results in an important phenomenon called delocalisation, which spreads out the MOs over the chain, allowing the electrons to move freely across it. It is actually possible to come up with an expression relating the length of the chain to the absorbed wavelength, which was done in my ILA. The result is that a molecule needs a conjugated system with at least five or six double bonds before it can absorb visible light, and hence we see why most molecules are in fact colourless.
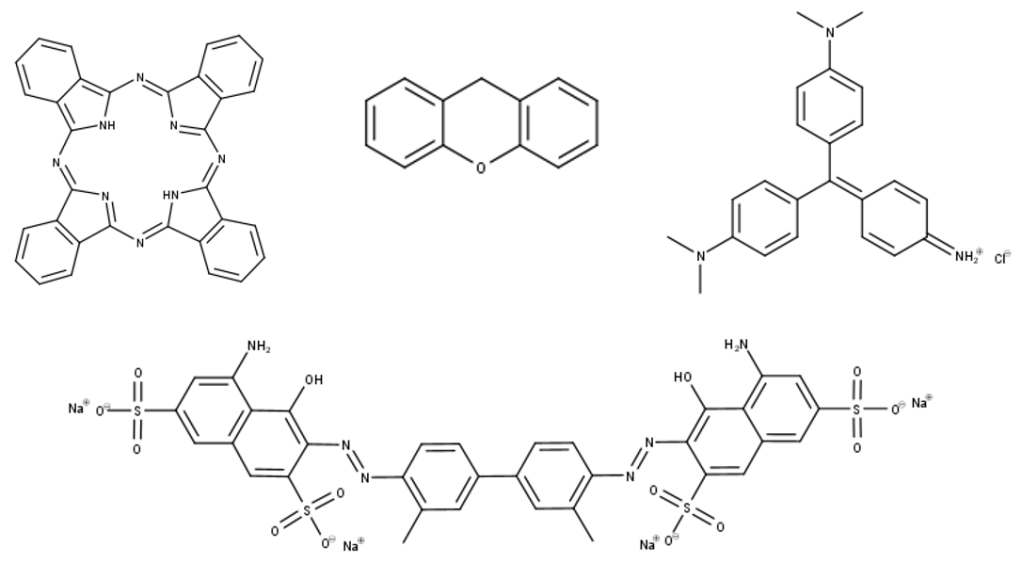
The synthesis of dyes requires an understanding of their structure. We already know that they must contain a conjugated system, and this part of the dye is called the chromophore. Any additional groups attached slightly alter the colour of the dye and are called auxochromes. The variety of structures this vary broad description allows is immense. A number of the chromophores explored in my ILA are shown below to show just how varied the dyes can be.
It is all well and good being able to create dyes, but they are useless if we have no way of attaching them to materials. Material structure is very diverse, but often the materials we dye have polymer structures. For example, plant fibres are composed of cellulose, a polymer of glucose. Animal fibres are made of proteins which are chains of amino acids. Even synthetic materials such as nylon are polymers. These polymers can stack together and align, creating a fibrous structure with pores running through it. The dye molecules are able to travel through these pores and attach themselves to the material through different kinds of bonding.
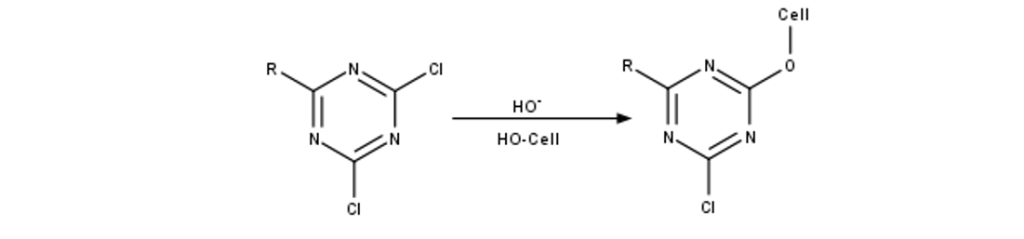
Direct dyes are the simplest to apply as these form strong enough interactions, often ionic or strong dispersion forces, to hold the dye in place without any other input. Another type of dyes called reactive dyes can quite literally react with the material, forming strong covalent bonds to it. An example of this is shown below.
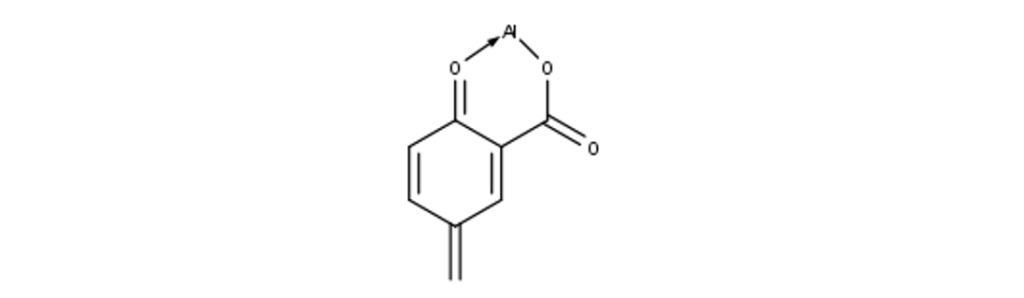
There are, however, situations when the dye cannot hold itself onto the material well enough, and a mordant must be used. Mordants are inorganic metal salts that help fix a dye to a material. The metal ion in the mordant forms something called a chelation with both the dye molecule and the material, acting as a bridge between them, holding them together. The structure of one of these chelations is shown here.
Dyes are not just useful in the chemical world; they occur all the time in nature too. Two interesting examples I covered in my ILA were retinal and chlorophyll. Retinal is able to harness its ability to absorb visible light to help us see colour by altering its structure when it absorbs light. Chlorophyll is able to use the energy it absorbs from visible light to help plants photosynthesise. So, it really is impossible to go anywhere without seeing, wearing, or using dyes in some shape or form and it is impossible to deny their importance in our world.
To view Alex’s full article, follow this link below.

